 Cadmiun, Cd, is to the left and below sulfur, so both electron shielding (down a group) and increase of the number of protons (down a period) lead to predict the cadmium has a larger atomic radius than sulfur. So far, you can rank the atomic radius of sulfur, chlorine, fluorine, and oxygen, in increasing order as: O < F < Cl < S, concluding that O, F, and Cl have smaller atomic radius than S. 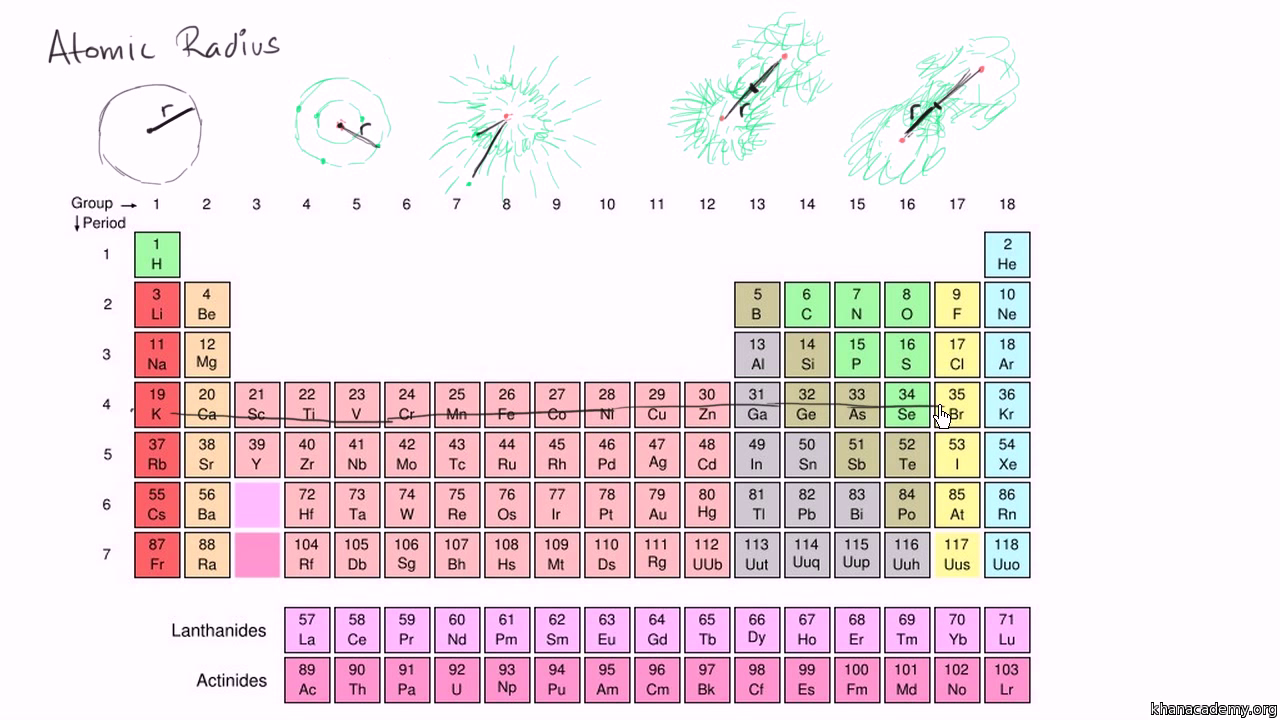
That permits you to compare the size of the elements in a group: Fluorine and chlorine are in the same group (17), with chlorine directly below fluorine, so the atomic radius of chlorine is larger than the atomic radius of fluorine.Sulfur and oxygen are in the same group (16), with sulfur directlly below oxygen, so sulfur the atomic radius of sulfur is larger than the atocmi radius of oxygen. 
Atomic radius increases from top to bottom within a group due to electron shielding. With that: Oxygen and fluorine are in the period 3, being oxygen to the left of fluorine, so oxygen is larger than fluorine.Sulfur and chlorine are in the period 4, being sulfur to the left of chlorine, so sulfur is larger than chlorine. Hence, you can compare the elements that belong to a same period and predict that the atom with lower atomic number (number of protons) will haver larger atomic radius. It is usually the mean or typical distance from the center of the nucleus to the boundary of the surrounding cloud of electrons.Įxplanation: Down a period, atomic radii decrease from left to right due to the increase in the number of protons and electrons across a period: when a proton is added the pull of the electrons towards the nucleus is larger, so the size of the atom decreases. For example, Fluorine has a smaller radius than Oxygen. 
Therefore, each electron is pulled stronger and the radius decreases. When you move right the row, the element has larger nuclear charge (more protons) for the same number of energy shells. Atomic radius of a chemical element is a measure of the size of its atoms. For example, Oxygen has a smaller radius than Sulfur. Also Read : PLEASE HELP AGAIN lol thank you :))Ītomic radius of sulfur is 88 pm Atomic radius of chlorine is 79 pmAtomic radius of cadmium is 161 pmAtomic radius of fluorine is 42 pmAtomic radius of oxygen is 48 pm. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
